 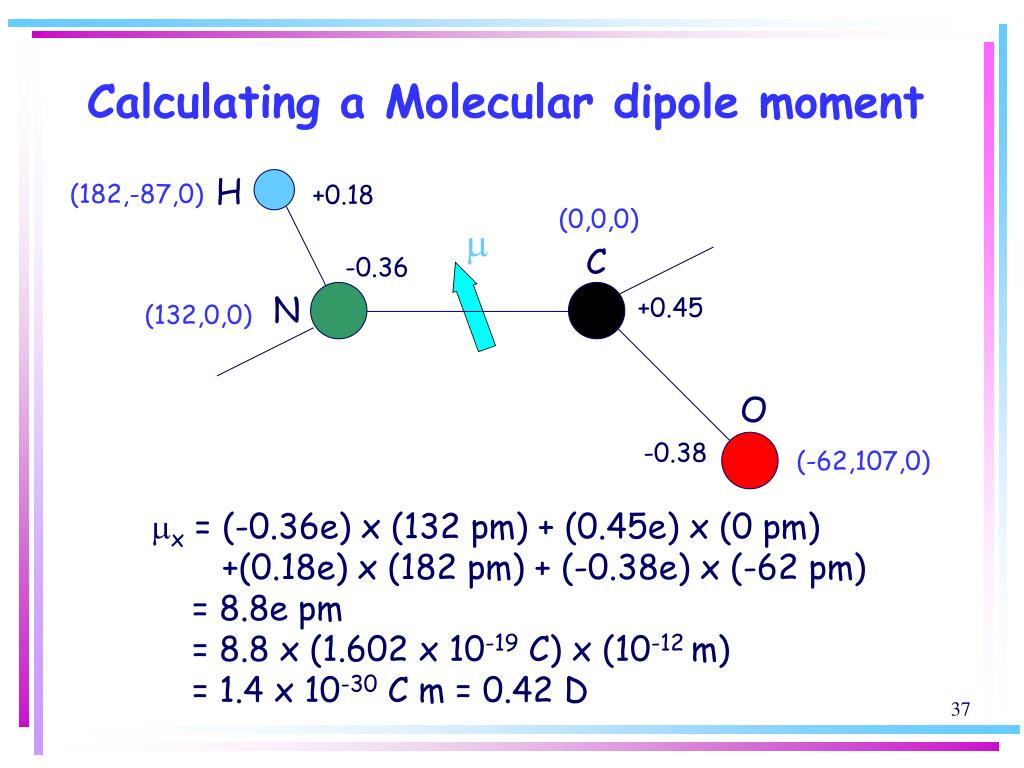
Considering the molar mass and hydrogen bonding, rank the hydrides from highest boiling point to lowest boiling point. For group 5A hydrides, first determine their relative molar mass, then determine whether each molecule exhibits hydrogen bonding. Thus, if the molecule shows hydrogen bonding, the boiling point of the substance will be higher than expected owing to the strength of the hydrogen bond relative to London dispersion forces. Because dipoles of the bond cancel each other. (i) (Refer to Image 1) (ii) (Refer to Image 2) (iii) (Refer to Image 3) (iv) (Refer to Image 4) Above molecules have dipole moment zero. This is due to the hydrogen bonding exhibited by the molecules of water. Dipole moment:- The polarity of covalent bond can be conveniently measured in terms of physical quantity called dipole moment. However, it is observed that the boiling point of H2O is highest among these hydrides. Which of the following pairs of molecules is listed in order of decreasing net dipole moment (from more polar molecule to less polar molecule) A. This is because, the electronegativity difference between the bonded atoms is the highest in N H3 (because the electronegativity of N is the highest among N, P, Sb and As atoms). In contrast, the dipole moments of amines decrease with substitution, starting with ammonia, which has a dipole moment of 1.47 D. So in PH 3, the small dipole moment of each P-H bond points from P + to H - (as shown below). The dipole moment of a polar covalent bond conventionally points from the positive center to the center of the negative charge. It occurs whenever positive and negative charges are separated. The dipole moment is expressed in a unit called Debye (D). Accurate values of the dipole moments ( v2 1 0.5740 0.0002 D) and of the transition frequencies have been obtained. The dipole moment of N H3 is the highest among the given molecules. The dipole moment is 0.58 D, which increases with substitution of methyl groups in the series: CH3PH2, 1.10 D (CH3)2PH, 1.23 D (CH3)3P, 1.19 D. The bond dipole moment is a measurement of the polarity of a chemical bond within a molecule that is based on the electric dipole moment concept. Among these hydrides, the molar mass increases in the following order: H2O < H2S < H2Se < H2Te Therefore, the boiling point of H2O is expected to be the lowest. Nine transitions belonging to the v4 and v4 bands of PH 3 have been brought in resonance with CO 2 laser lines, using an intracavity Stark cell. C) The electrons in a polar bond are found nearer to the more electronegative element. B) Dipole moments result from the unequal distribution of electrons in a molecule. Observed dipole moment components (a 1.513(6) D, b 0 D, c 1.259(3) D. For example, consider group 6A hydrides: H2O, H2S, H2Se, and H2Te. 3.4 (12 reviews) Which of the following statements is incorrect A) Ionic bonding results from the transfer of electrons from one atom to another. High order dipole moment surfaces of PH3 and ab initio intensity predictions in the Octad range. Consequently, the boiling point will also be higher. The greater the molar mass, the greater the strength of the London dispersion forces (a type of intermolecular force of attraction between two molecules). Last, a greater magnitude of dipole will cause stronger attraction.Step 1: To rank the molecules from highest boiling point to lowest boiling point, first consider the molar mass of the molecules. An ion with higher charge will make the attraction stronger. Therefore there will be dipole dipole interactions. 
The closer ion and polar molecule are, the stronger the intermolecular force is between polar molecule and ion. Due to back bonding in PF 3 it have greater dipole moment than PH 3. The strength of ion-dipole interaction is based on the distance between ion and polar molecule, the charge of the ion, and dipole magnitude. \) is the distance between 2 separated charges 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
